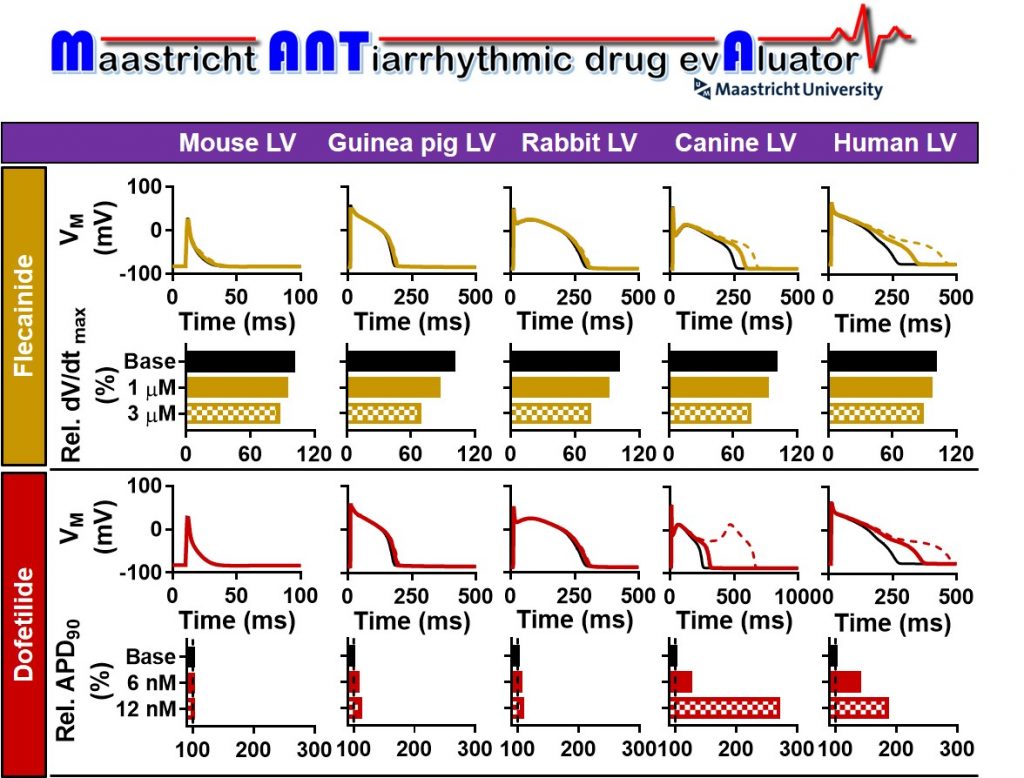
We are looking for a highly motivated PhD candidate to join a multidisciplinary team developing patient-level models for simulating virtual clinical trials in patients with atrial fibrillation, the most common heart rhythm disorder! In this 4-year project, the PhD-candidate will join an interdisciplinary research team involving clinicians and scientists from the Depts of Cardiology, Physiology and Biochemistry at the CARIM School for Cardiovascular Diseases, Maastricht University aiming to develop a highly innovative virtual-patient model that can simulate the clinical state (including the absence or presence of AF) during the entire lifetime of a cohort of thousands of virtual patients with minute-level resolution.
The ideal candidate has successfully completed a master’s degree (or equivalent) in technical medicine, medical engineering, medicine, biomedical engineering or related disciplines. He/she has a strong intrinsic motivation to commit to an interdisciplinary 4 year PhD-program integrating computational approaches and complex clinical data. He/she also has a demonstrable interest in cardiovascular research, and an analytical mindset. Advanced knowledge of scientific English and excellent communication skills are required. Previous experience with programming and/or computational modeling, experience with clinical trials, or prior research experience in (cardiovascular) epidemiology or atrial fibrillation is considered a plus.
Fore more information, and to apply, see the advert on AcademicTransfer: https://www.academictransfer.com/en/297262/phd-student-promovendus-in-computational-modeling-of-atrial-fibrillation-fhmlschool-for-cardiovascular-diseasescarim-cardiology/
 The Dutch Research Council (NWO) has awarded a prestigious Vidi grant worth 800,000 euros to Dr. Jordi Heijman. Dr. Heijman is one of three Vidi laureates from Maastricht University in 2020 (out of a total of 81 awardees selected from 503 eligible proposals) with a proposal entitled ‘Time traveling to treat heart rhythm disorders’.
The Dutch Research Council (NWO) has awarded a prestigious Vidi grant worth 800,000 euros to Dr. Jordi Heijman. Dr. Heijman is one of three Vidi laureates from Maastricht University in 2020 (out of a total of 81 awardees selected from 503 eligible proposals) with a proposal entitled ‘Time traveling to treat heart rhythm disorders’. During this year’s elections of the European Working Group on Cardiac Cellular Electrophysiology (EWGCCE), Dr. Jordi Heijman has been elected as incoming chair-elect of the working group. Jordi was elected as nucleus member of EWGCCE in June 2018 and has been the working group’s secretary since October 2018. He is currently also co-chairing a position paper on “Relevance, Opportunities and Limitations of Experimental Models for Cardiac Electrophysiology Research” together with Prof. Katja Odening from Bern. Jordi has started his position as chair-elect and treasurer of the EWGCCE in September of this year, working closely together with the current chairperson, Prof. Ana Maria Gomez from Paris. He will become chairman of the EWGCCE for the 2022-2024 term. As chairman, Jordi aims to strengthen interdisciplinary collaborations and education in cardiac cellular electrophysiology, among other things.
During this year’s elections of the European Working Group on Cardiac Cellular Electrophysiology (EWGCCE), Dr. Jordi Heijman has been elected as incoming chair-elect of the working group. Jordi was elected as nucleus member of EWGCCE in June 2018 and has been the working group’s secretary since October 2018. He is currently also co-chairing a position paper on “Relevance, Opportunities and Limitations of Experimental Models for Cardiac Electrophysiology Research” together with Prof. Katja Odening from Bern. Jordi has started his position as chair-elect and treasurer of the EWGCCE in September of this year, working closely together with the current chairperson, Prof. Ana Maria Gomez from Paris. He will become chairman of the EWGCCE for the 2022-2024 term. As chairman, Jordi aims to strengthen interdisciplinary collaborations and education in cardiac cellular electrophysiology, among other things. After defending his Master Thesis research via Zoom with a presentation on “Assessing Outcomes and Risk Factors Of Atrial Fibrillation Through A Lifetime Population-Level Markov Model“, Cristian Barrios Espinosa officially graduated from the Master Systems Biology program on July 6th during an online ceremony. Cristian received an 8.8 out of 10 for his master thesis research and graduated cum laude. Congratulations! From September 2020 onwards, Cristian will be working as a PhD student at the Karlsruhe Institute of Technology, Germany, as part of the Marie-Curie ITN project
After defending his Master Thesis research via Zoom with a presentation on “Assessing Outcomes and Risk Factors Of Atrial Fibrillation Through A Lifetime Population-Level Markov Model“, Cristian Barrios Espinosa officially graduated from the Master Systems Biology program on July 6th during an online ceremony. Cristian received an 8.8 out of 10 for his master thesis research and graduated cum laude. Congratulations! From September 2020 onwards, Cristian will be working as a PhD student at the Karlsruhe Institute of Technology, Germany, as part of the Marie-Curie ITN project  During the 2019-2020 academic year,
During the 2019-2020 academic year,