During the summer, Su Min Pack, a bachelor student of the Biomedical Sciences program at Maastricht University, will join the team to obtain experience doing cardiovascular research. Su Min will help to integrate a recently published model of the human induced pluripotent stem cell (hIPSC)-derived cardiomyocyte into our Maastricht Antiarrhythmic Drug Evaluator (MANTA) tool, thereby facilitating comparisons between the electrophysiological response of hIPSC-derived cardiomyocytes and adult human cardiomyocytes to different drugs.
During the summer, Su Min Pack, a bachelor student of the Biomedical Sciences program at Maastricht University, will join the team to obtain experience doing cardiovascular research. Su Min will help to integrate a recently published model of the human induced pluripotent stem cell (hIPSC)-derived cardiomyocyte into our Maastricht Antiarrhythmic Drug Evaluator (MANTA) tool, thereby facilitating comparisons between the electrophysiological response of hIPSC-derived cardiomyocytes and adult human cardiomyocytes to different drugs.
Graduation Lian Laudy
On July 11, 2019, Lian Laudy successfully completed her Masters with the official graduation ceremony of the Master Systems Biology of Maastricht University. She received an excellent grade (8.9/10) for her Master thesis research “Computational modelling of calcium-dependent signalling pathways and their long-term effects on cardiac calcium handling and arrhythmogenesis” and graduated cum laude – congratulations! Since then, Lian has started working as a PhD student in the team of Dr. Joost Lumens at the Department of Biomedical Engineering on a joint project and will continue to be involved in the lab.

Updated publications
We have finally updated our list of publications, which was highly overdue since a number of publications have been accepted recently. These new papers include an extensive review on post-operative atrial fibrillation published in Nat Rev Cardiol and a general description about the role of calcium in the heart aimed at young aspiring scientists published in Frontiers for Young Minds.

Panel Discussion at Gordon Research Seminar
Last week, Jordi Heijman participated in the panel discussion ‘How to build your own lab’ together with Profs. Patrick Boyle, Ursula Ravens and Crystal Ripplinger during the 2019 edition of the Gordon Research Seminar on Cardiac Arrhythmia Mechanisms, which took place in Lucca (Barga) , Italy. During the lively discussion, a wide range of topics, including obtaining faculty positions, setting up a lab, and maintaining a reasonable work-life blend were addressed. The discussion was moderated by Drs. Axel Loewe and Mathilde Rivaud, chairs of this edition of the Gordon Research Seminar.

New PhD Position in Computational Modeling at Maastricht University
We have the great honor to collaborate with Dr. Joost Lumens (Dept. of Biomedical Engineering, Maastricht University) on his 2018 ERA-CVD project EMPATHY (“Electromechanical Presages of Sudden Cardiac Death in the Young: integrating imaging, modelling and genetics for patient stratification”). For this project we are looking for a highly motivated PhD candidate to join our multidisciplinary and translational research team, who is eager to be challenged and passionate to use computational modeling to solve the complex puzzle of sudden cardiac death in the young. In particular, the PhD student will develop and validate a fully coupled, multi-scale computational model of cardiac electro-mechanics. This model will link the dynamics of cellular electrophysiology with cardiac mechanics and circulatory hemodynamics. This unique tool will then be used to study electro-mechanical coupling across scales (i.e. cell, tissue, organ and circulation) during exercise in the normal and the diseased heart.
More information about the project and how to apply can be found on Academic Transfer.
Happy Holidays!
The Heijman lab wishes you and your loved ones happy holidays and a happy, healthy and successful 2019! We are looking forward to continuing the existing collaborations and start many new ones in the year to come!
Several new publications
During the last weeks a number of new publications co-authored by members of the team have been published, including 1) a state-of-the-art paper about the impact of molecular mechanisms for cardiac arrhythmia management from the Working Group on Cardiac Cellular Electrophysiology of the German Cardiac Society, 2) an editorial about the potential antiarrhythmic effects of resveratrol, and two original research articles: 3) one paper on predicting the functional effects of genetic variants in SCN5A using machine-learning approaches and 4) one paper about atrial profibrotic, electrical and calcium-handling remodeling in heart failure patients with and without atrial fibrillation.
A full overview of all publications can be found here.
Computing in Cardiology and Frontiers in Computational Electrocardiology 2018
Last week Jordi Heijman co-organized the 45th annual Computing in Cardiology conference, which attracted almost 400 guests from >50 different countries. The conference started on Sunday, September 23rd with a hands-on workshop using 4 different simulation tools employing the computer facilities of Maastricht University. From Monday until Wednesday, the conference took place at the MECC Conference Center with four parallel sessions with oral presentations and two poster sessions of >70 posters each. Besides high-quality science, participants enjoyed an extensive social program, including a lunch cruise and various activities on Monday afternoon, a visit to the Bonnefantenmuseum and dinner at Ipanema on Monday evening, and a visit to Maastricht city hall on Tuesday evening.

After Computing in Cardiology 2018, we also organized the 5th edition of Frontiers in Computational Electrocardiology (FiCE) 2018, this time as a satellite symposium immediately after CinC. The FiCE symposium focused on the translation of computer simulation and image integration to clinical practice and included key-note lectures by Prof. Natalia Trayanova (Johns Hopkins, Baltimore, USA) and Fu-Siong Ng (Imperial College, London, UK).
Best oral communication award at Early Career Physiologists’ Symposium – Europhysiology 2018 for Henry Sutanto
London, 13-16th September 2018: Henry Sutanto presented his project entitled “Subcellular organization of ryanodine receptors and Ca2+ channels modulate the propensity of spontaneous Ca2+ waves and cardiac arrhythmias”, which has recently been published in Frontiers in Physiology, as both oral communication at the Early Career Physiologists’ Symposium 2018 and poster presentation at Europhysiology 2018. In this biennial event organized by The Physiological Society (Physoc), Deutsche Fysiologie Gesellschaft (DFG), Scandinavian Physiological Society (SPS) and Federation of European Physiological Society (FEPS), Henry Sutanto was awarded the best oral presentation after presenting his project in front of more than 100 early career physiologists.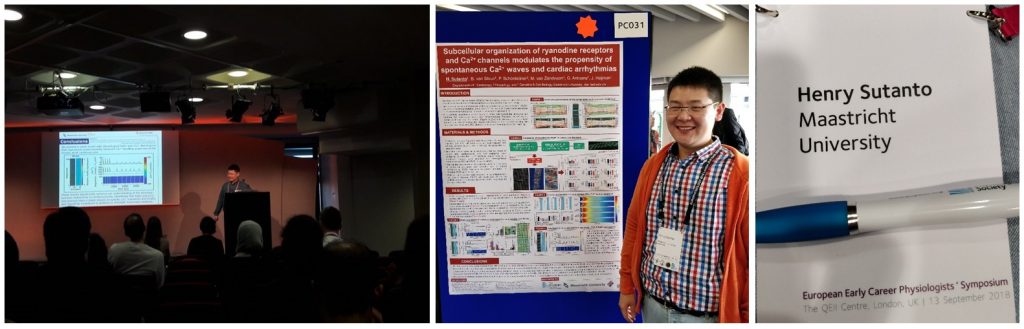
Lab Get-Together 2018

On September 5, the team came together for the annual lab get-together to enjoy some pizza and drinks to celebrate the excellent results achieved so far this year!